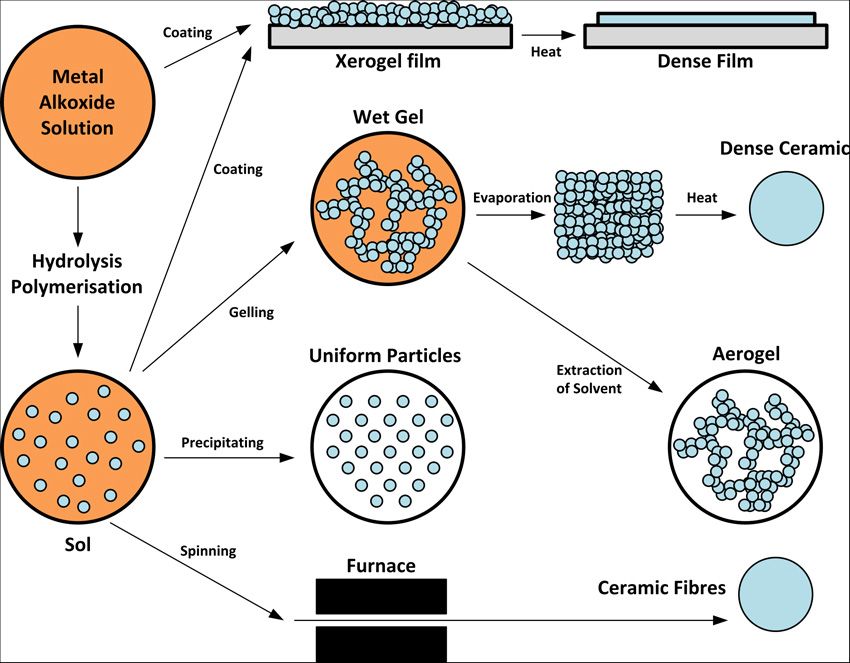
The physico-chemical process underlying digestion is called Ostwald ripening. It results in cleaner and bigger particles. Similar processes are often used to separate chemically similar elements, such as the rare earth metals.ĭigestion, or precipitate ageing, happens when a freshly-formed precipitate is left, usually at a higher temperature, in the solution from which it is precipitated. To identify the cation, the color of the precipitate and its solubility in excess are noted. To do this, an alkali first reacts with the unknown salt to produce a precipitate which is the hydroxide of the unknown salt. Precipitate formation is useful in the detection of the type of cation in salt. This simplifies the above equations to the following: A precipitation reaction is a special kind of double-replacement reaction where two soluble compounds react to form an insoluble solid called a precipitate. 3 In case of an inorganic chemical reaction leading to precipitation, the chemical reagent causing the solid to form is called the precipitant. 1 2 The solid formed is called the precipitate. In this case, any spectator ions (those which do not contribute to the reaction) are left out of the formula completely. Precipitate product definition Precipitate tests - Chemical analysis - (CCEA) - BBC SpletPrecipitation (Chemical) Chemical precipitation is the process of. In an aqueous solution, precipitation is the process of transforming a dissolved substance into an insoluble solid from a supersaturated solution. This reaction can be written emphasizing the dissociated ions in a combined solutionĪg +(aq) + NO 3 -(aq) + K +(aq) + Cl -(aq) → AgCl(solid) + K +(aq) + NO 3 -(aq)Ī final way to represent a precipitate reaction is known as a net ionic reaction. The silver chloride(AgCl) has formed a solid, which is observed as a precipitate. Fehling's solution is blue due to dissolved Cu 2+. Synonyms for PRECIPITATE: hurried, rushed, rash, precipitous, sudden, impulsive, hasty, headlong Antonyms of PRECIPITATE: deliberate, calculated, prolonged. In meteorology a precipitate is liquid or solid water (rain, snow, etc.) falling from the sky. (Zumdahl, 2005)ĪgNO 3(aq) + KCl(aq) → AgCl(s) + KNO 3(aq) Precipitate: In chemistry, a solid formed by a change in a solution, often due to a chemical reaction or change in temperature that decreases solubility of a solid. precipitateprecipitate2 /prsptt/ noun countable technicalHC a solid substance that has been chemically separated from a liquidExamples from the. Reagent oxidation is a special case of reagent coagulation in which oxidising reagents (e.g. If this energy is not available, and no suitable nucleation surface is available, supersaturation occurs.Īn example of a precipitation reaction: Aqueous silver nitrate (AgNO 3) is added to a solution containing potassium chloride (KCl) and the precipitation of a white solid, silver chloride is observed. Chemical precipitation or reagent coagulation precipitates impurities from purified water via change of pH, electrooxidising potential or coprecipitation using precipitating agents (coagulants) such as ferrous or aluminium sulphates (IAEA, 1992). 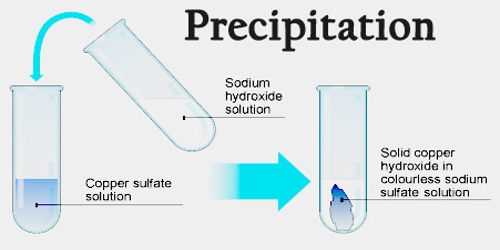
The creation of a hypothetical solid particle includes the formation of an interface, which requires some energy based on the relative surface energy of the solid and the solution. 
Because equilibrium constants of this kind are written as products, the resulting K 's are commonly known as solubility products, denoted by Ks or Ksp. Precipitation from a solid solution is also a useful way to strengthen alloys this process is known as solid solution strengthening.Īn important stage of the precipitation process is the onset of nucleation. Quantitative Chemistry Gravimetric Analysis: PRECIPITATE Double Displacement Reaction Definition and Examples WebA precipitation reaction is a reaction that. But because solid substances do not normally appear in equilibrium expressions, the equilibrium constant for this process is. This effect is useful in many industrial and scientific applications whereby a chemical reaction may produce a solid that can be collected from the solution by various methods (e.g. Precipitation reactions can be used for making pigments, removing salts from water in water treatment, and for qualitative chemical analysis. 3 Representation using chemical equations Related topics: Chemistry precipitate precipitate 2 / prsptt / noun countable technical HC a solid substance that has been chemically separated from a liquid Examples from the Corpus precipitate I producing fixed capital, there would be a precipitate decline in those sectors.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
